August 11, 2021
Schlafender Hase Team
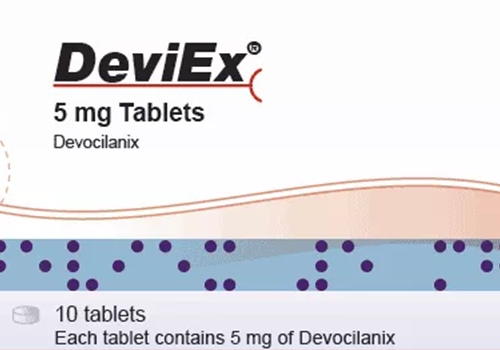
Make Sure Your Barcode Makes the Grade
In this interview, Mike Baird talks about TVT Barcode and what TVT customers can expect from this new module and the newly released version 10.2 of TVT, the Text Verification Tool.